Part
01
of sixteen
Part
01
What is Helium-3 and why it is so important?
Key Takeaways
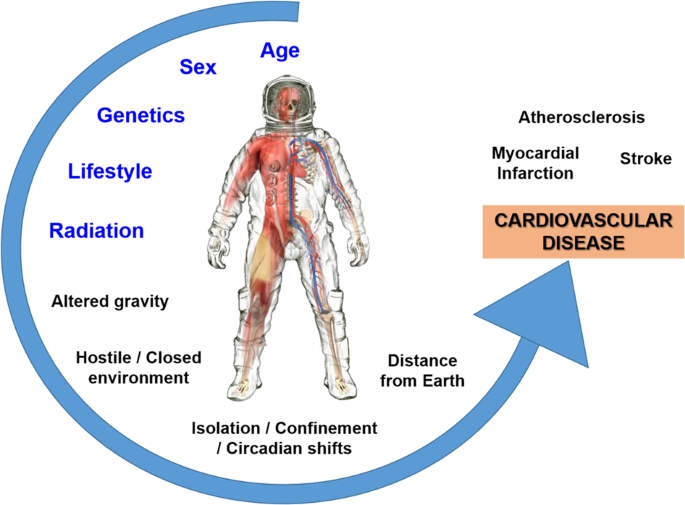
- Helium-3 (abbreviated as 3He) is an isotope of Helium (abbreviated as He), the second element in the periodic table and a noble gas.
- The resource is currently "most sought-after" for its potential use as a fuel for nuclear fusion, a safe form of nuclear energy that is not radioactive and avoids dangerous waste products.
- Helium-3 has other valuable applications, such as cryogenics, due to its ability to lower temperatures to close to absolute zero, non-radioactive medical imaging, nuclear weapon detection, quantum computing, and neutron research.
- Although Helium-3 is "virtually nonexistent here on Earth," where it is estimated that only .01 metric tons currently exist, the Earth's moon is believed to be home to 1,100,000 metric tonnes of the resource, representing "enough to power human energy needs for up to 10,000 years."
Introduction
The research team has curated a detailed overview of Helium-3, comprised of a definition of the isotope, its importance and/or value to humans, and the current motivation for considering the moon as a source of Helium-3. The full details of this analysis have been provided directly below.
I. Helium-3 Definition
- Helium-3 (abbreviated as 3He) is a gas derivative of the element Helium (abbreviated as He), the second element in the periodic table and a noble gas.
- Unlike Helium, which is defined by its "2 protons, 2 neutrons, and 2 electrons," Helium-3 has only one neutron and is, therefore, a light isotope of Helium.
- The substance was first theorized to exist in 1934 by Mark Oliphant, an Australian nuclear physicist, who assumed that Helium-3 would be a radioactive isotope. This myth was debunked after natural samples of Helium-3 were subsequently discovered from the atmosphere as well as gas wells.
- Notably, Helium-3 is the "only stable isotope of any element with more protons than neutrons" apart from Hydrogen-1.
II. Importance of Helium-3
- Helium-3 is currently "most sought-after" for its potential use as an energy source, according to a preponderance of experts including space organizations (e.g., European Space Agency), educational institutions (e.g., Institut Polytechnique De Paris), energy trades (e.g., Energy Industry Review) and Helium-3 futurists (e.g., Christopher Barnatt).
- Specifically, it has been believed since 1988 that the isotope could be used as a fuel for nuclear fusion, a safer form of nuclear energy that is not radioactive and avoids dangerous waste outputs. At present, nuclear power plants depend on the separate process of nuclear fission, which releases radioactive waste alongside any energy gains.
- Scientists have theorized that Helium-3 and Deuterium could serve as fuels for "aneutronic" fusion reactors, wherein the two would be fused together to create Helium and a proton, thereby generating a "highly efficient form of nuclear power with virtually no waste and no radiation."
- Currently, the University of Wisconsin-Madison is testing a small reactor of this structure at its Fusion Technology Institute. However, no trials have resulted in net power output. Although "a number of other types of reactors have been proposed" for Helium-3 nuclear fusion, most remain theoretical and appear to require more energy to operate than they ultimately produce.
- This early developmental state of Helium-3 nuclear fusion technology, however, is considered "almost irrelevant at this point...[as] no-one can dismiss the potential of... Helium-3 power generation."
- Moreover, Helium-3 has other valuable applications such as cryogenics due to its ability to lower temperatures to close to absolute zero, non-radioactive medical imaging, nuclear weapon detection, quantum computing, and neutron research.
III. The Moon as a Source of Helium-3
- Considering the importance of Helium-3, governments, companies, and other institutions are evaluating the moon as a primary future source of this gas.
- Seeking out this extra-terrestrial extraction location is viewed as necessary, given that Helium-3 is "virtually nonexistent here on Earth." This is because the planet's atmosphere prevents any Helium-3 from arriving.
- It is estimated that only .01 metric tons of Helium-3 exist on Earth. Although recent research indicates that Helium-3 quantities on the planet may be higher than previously thought, the available quantities remain very low.
- Additionally, while Helium-3 can be generated as a by-product of nuclear weapons, this could yield only 15 kilos per annum given current capabilities.
- In contrast, the Earth's moon lacks an atmosphere and has therefore been exposed to solar winds with Helium-3 for "billions of years."
- As a result, it is widely estimated that the planet's satellite is home to 1,100,000 metric tonnes of Helium-3 within the first few meters of its surface, making it an abundant source of the resource.
- Such volume would be "enough to power human energy needs for up to 10,000 years," while just a fully loaded Space Shuttle of Helium-3 from the moon could power the United States for a full year.
- Meanwhile, it is worth noting that Jupiter is separately being evaluated as a source for mining Helium-3, owing to the planet's even more vast quantities of the isotope. However, transportation from a more distant location would add complexity.
Research Strategy
For this research on Helium-3, the research team leveraged the most reputable sources available in the public domain, including space organizations (e.g., European Space Agency), educational institutions and resources (e.g., Institut Polytechnique De Paris, EDinformatics), space and technology trades (e.g., Energy Industry Review, Inverse) and Helium-3 experts (e.g., Christopher Barnatt).